Hence, neoadjuvant immunotherapy or neoadjuvant targeted treatment seems to be a promising treatment for operable locally advanced NSCLC. We designed this clinical trial aiming to investigate the efficacy of different neoadjuvant therapies (neoadjuvant immunotherapy with(out) chemotherapy, neoadjuvant chemotherapy, and neoadjuvant targeted therapy) in operable locally advanced NSCLC. gemcitabine plus cisplatin (GC chemotherapy) as neoadjuvant therapy) also achieved better results in patients with locally advanced epidermal growth factor receptor (EGFR) mutation-positive NSCLC ( 10). In addition to neoadjuvant immunotherapy, the EMERGING-CTONG 1103 trial (erlotinib vs. 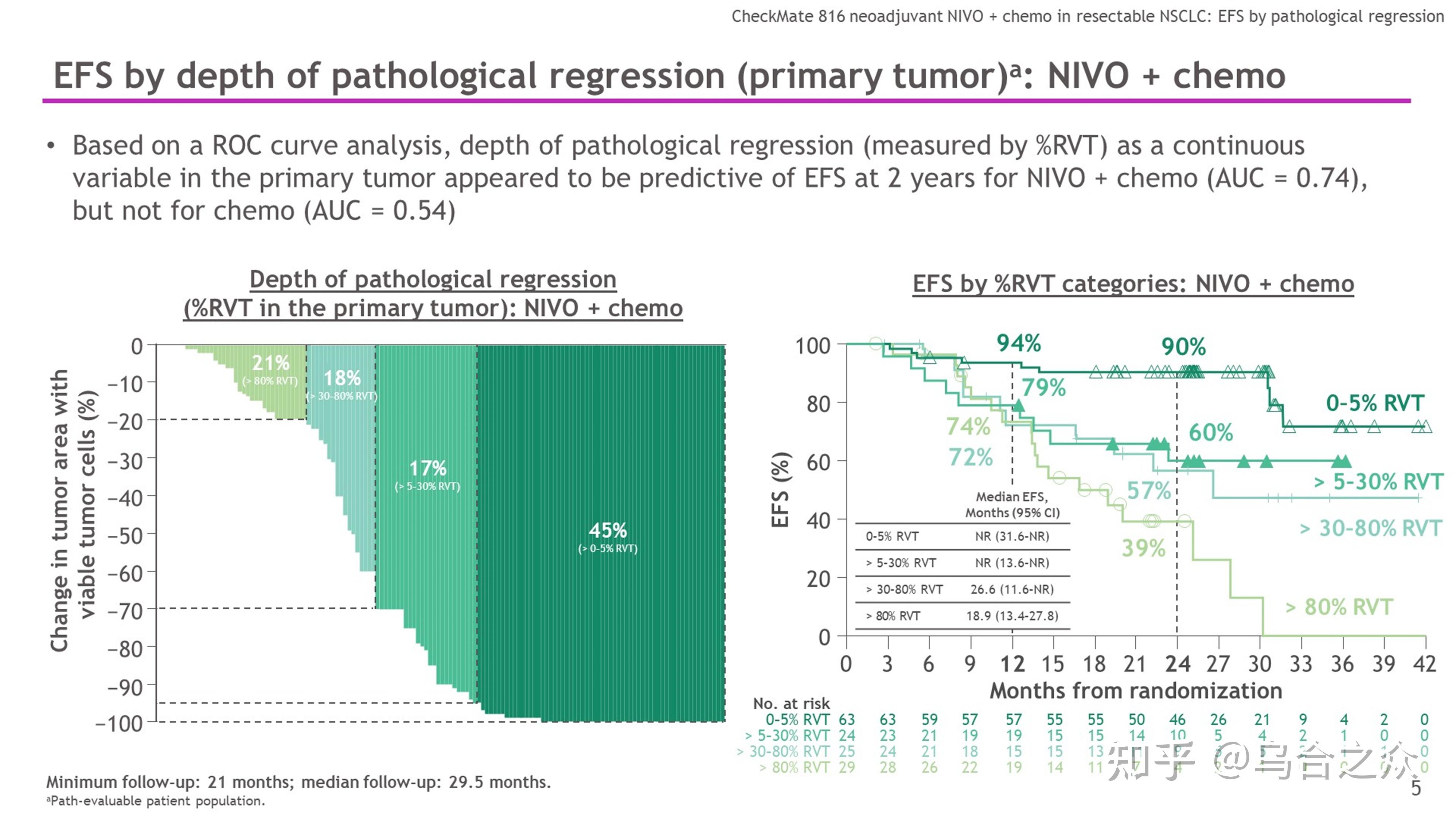
Then, the NADIM trial (neoadjuvant chemotherapy plus nivolumab) and NEOSTAR trial (neoadjuvant nivolumab or nivolumab plus ipilimumab) also demonstrated the potential value of neoadjuvant immunotherapy in operable NSCLC ( 8, 9). Checkmate 159 was the first clinical trial to report neoadjuvant immunotherapy before surgery in 21 limited-stage NSCLC patients who received 2 cycles of nivolumab ( 7). The emergence of immune checkpoint inhibitors (ICIs), including programmed cell death 1 (PD-1) or programmed cell death ligand 1 (PD-L1) antibodies and cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) antibodies, has completely revolutionized the situation of neoadjuvant chemotherapy for operable locally advanced NSCLC. Based on this unsatisfactory benefit, researchers have focused on exploring various neoadjuvant therapies in operable locally advanced NSCLC. Although neoadjuvant chemotherapy represents a promising treatment strategy that significantly improves the survival rate in operable locally advanced NSCLC, the 5-year survival rate remains less than 50% in these patients ( 5, 6). For locally advanced NSCLC, the standard treatment is multidisciplinary therapy, including neoadjuvant therapy, complete surgical resection, and adjuvant treatment ( 3, 4). Of lung cancer cases, approximately 80% are classified as non-small cell lung cancer (NSCLC), and approximately one-third of NSCLC cases are diagnosed at a locally advanced stage ( 1, 2). Lung cancer is still the leading cause of cancer death in 2020 worldwide. Curative effect (pCR + MPR) was significantly better with neoadjuvant immunotherapy (P = 0.006, 95% confidence interval, 0.008–0.012). Different neoadjuvant therapies had a statistically significant effect on postoperative pathological tumor downstaging ( P = 0.017).Ĭonclusions: Neoadjuvant immunotherapy was associated with a trend toward better pCR than the neoadjuvant chemotherapy arm and neoadjuvant targeted therapy. 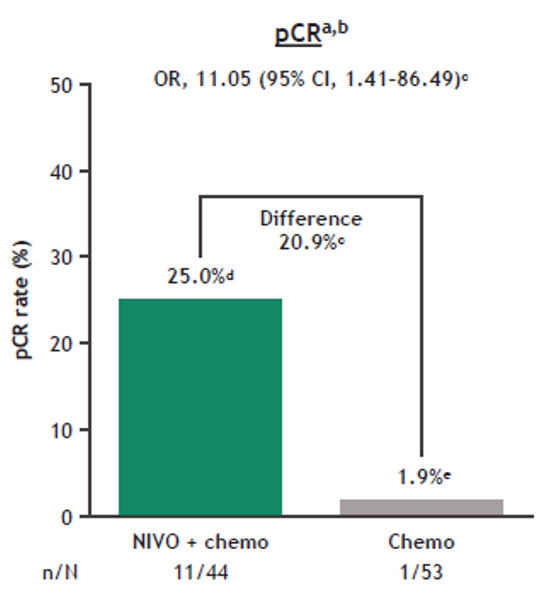
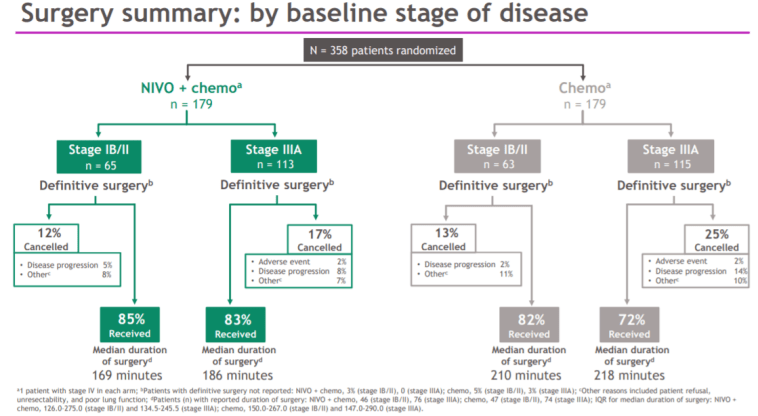
More importantly, we found that the curative effect of the neoadjuvant immunotherapy arm in pCR+MPR was better than that of the neoadjuvant chemotherapy arm and neoadjuvant targeted therapy arm. Results: An improved pathologic complete response was achieved in the neoadjuvant immunotherapy arm compared with the neoadjuvant chemotherapy arm and neoadjuvant targeted therapy arm. Major pathological response (MPR) and tumor regression rate (TRR) were also evaluated. Pathologic complete response (pCR) was evaluated as the primary endpoint. All 49 patients had surgical resection within 4–6 weeks after 2–3 cycles of neoadjuvant treatment consisting of immunotherapy (24 patients), chemotherapy (16 patients), and a targeted therapy (9 patients) regimen starting on the first day of each 21-day cycle. Methods: This study was conducted at Shanghai Chest Hospital and included eligible NSCLC patients who were 18 years old and had clinical stage IIB–IIIB disease. We report an interim analysis of 49 of 53 evaluable patients.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
